Prussian Blue Stain
Prussian Blue Stain Service: Detect Ferric Iron with Clarity and Confidence
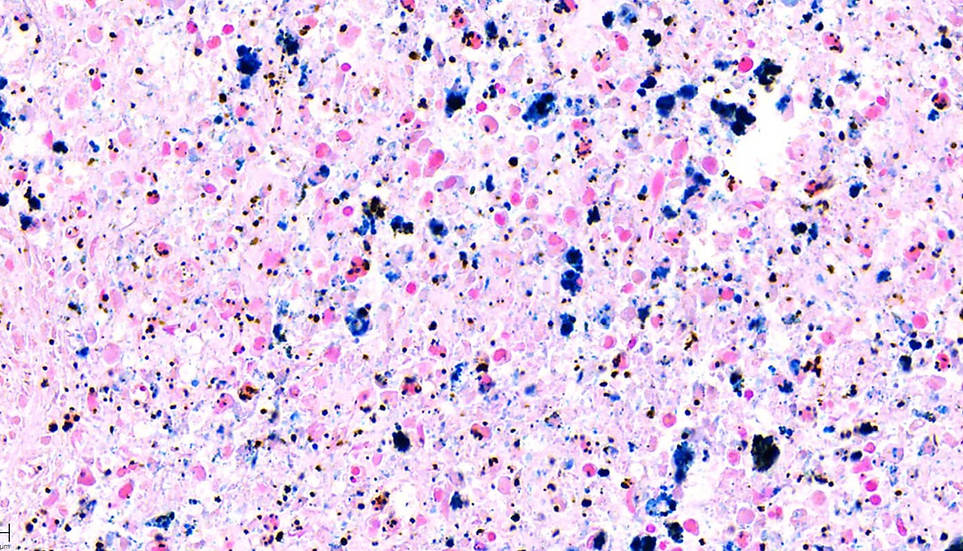
At iHisto, we offer Prussian Blue staining to detect ferric (Fe³⁺) iron in tissue sections with precision and clarity. This classic technique is essential for evaluating iron overload, hemosiderin deposition, and post-hemorrhagic changes in both clinical and preclinical samples.
🧪 Overview
The Prussian Blue stain identifies ferric iron (usually stored as hemosiderin) through a chemical reaction that produces deep blue iron deposits. At iHisto, we apply a carefully calibrated protocol to deliver high-contrast results — ideal for:
Hemochromatosis or hemosiderosis diagnosis
Iron-related toxicity screening
Microhemorrhage and trauma detection
Iron accumulation mapping in organ systems
We routinely process liver, spleen, kidney, bone marrow, and CNS tissues with excellent staining reproducibility.
⚙️ How Prussian Blue Staining Works
Ferric iron reacts with potassium ferrocyanide in an acidic environment to form insoluble ferric ferrocyanide (Prussian Blue) at sites of iron deposition.
Color Results:
Ferric iron (hemosiderin) → deep blue
Nuclei → red or purple (via nuclear fast red or eosin)
Background → pink
This distinct color separation enables easy identification of iron deposits in tissue sections.
🧬 Step-by-Step Staining Process
Fixation & Sectioning – FFPE tissue cut at 4–5 µm
Deparaffinization & Hydration – Slides cleared and hydrated into water
Iron Reaction – Incubated with potassium ferrocyanide and hydrochloric acid
Counterstaining – Nuclear fast red or eosin applied for contrast
Dehydration & Coverslipping – Prepared for brightfield microscopy and archiving
🔬 Applications of Prussian Blue Staining
This stain is widely used across human and animal tissues to evaluate iron metabolism and bleeding.
Common use cases:
Hemochromatosis and hemosiderosis – Iron overload in liver and spleen
Kidney and brain pathology – Post-hemorrhagic iron deposits
Toxicologic pathology – Iron accumulation in exposure studies
Bone marrow and spleen – Transfusion-related iron storage
CNS trauma research – Microhemorrhage and neuroinflammation
✅ Why Choose iHisto for Prussian Blue
✅ Crisp, selective detection of ferric iron
✅ Validated for rodent, primate, and human tissue
✅ Optional pairing with reticulin, trichrome, or H&E
✅ Whole-slide imaging with HistoCloud delivery
✅ Pathologist review and quantification available
We serve hospitals, research institutions, CROs, and biotech teams with tailored iron staining protocols and fast turnaround.
❓ FAQs
What does the Prussian Blue stain detect?
It detects ferric iron (Fe³⁺) in tissues, particularly in the form of hemosiderin.Which tissues are typically stained?
Liver, spleen, kidney, bone marrow, and CNS are most common, especially in iron metabolism studies.Can I receive digital versions of my slides?
Yes — we provide high-resolution whole-slide scans with secure cloud access or encrypted export.Can it be combined with other stains?
Yes — commonly paired with reticulin or trichrome for evaluating iron-fibrosis correlation or architectural context.
📩 Request a Quote or Consultation
Need reliable visualization of iron in tissue? iHisto’s Prussian Blue staining service delivers precise detection of ferric iron — whether you're studying metabolic disease, trauma, or therapeutic impact.
👉 Request a Quote or email info@ihist




